FDA Approves First Dual Therapy for Acute and Preventive Treatment of Migraine

Rimegepant is demonstrated for grown-up patients with verbose headaches.
Authorities with the FDA have supported rimegepant 75 mg (Nurtec ODT; Biohaven Pharmaceutical Holding Company Ltd.) for double treatment of intense headaches and the precaution therapy of headaches in grown-ups. As indicated by Biohaven, the medication item is the principal prescription supported for the two employments.
An oral calcitonin quality related peptide enemy, rimegepant is demonstrated for patients with wordy headaches. The medication was recently endorsed by the FDA for intense headaches with or without air in grown-ups.
As per Biohaven, around 95% of all patients with headache in the United States experience under 15 migraine days out of each month.
The new endorsed item mark permits utilization of rimegepant for up to 18 portions each month, which remembers treatment for both intense and preventive treatment for a similar patient.
BIOHAVEN told FDA About the Treatment
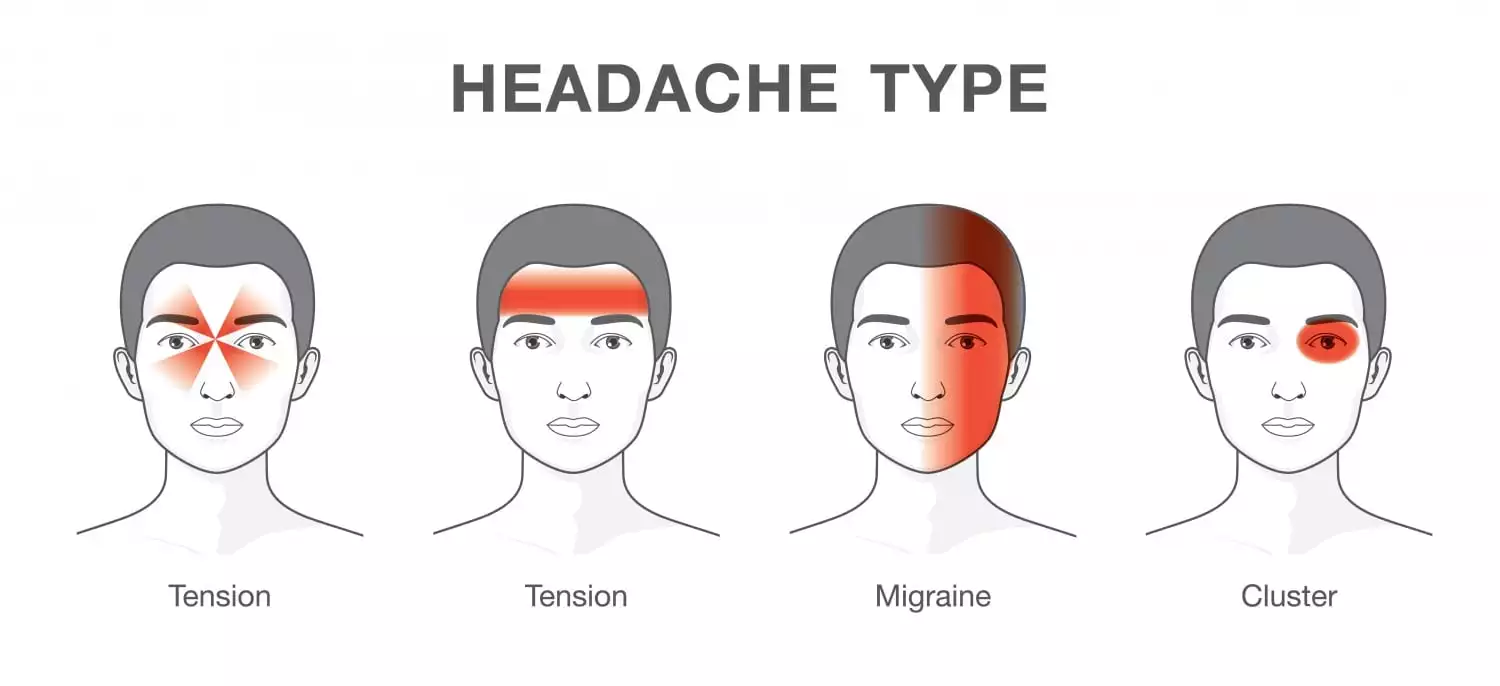
As indicated by Biohaven, headaches can be incapacitating, enduring 4 to 72 hours with numerous manifestations, including throbbing migraines of moderate to serious torment force that can be related with queasiness or regurgitating, affectability to sound, and affectability to light.
In a pre-arranged explanation, Peter J. Goadsby, MD, PhD, DSc, teacher of nervous system science at the University of California, Los Angeles and King’s College London, beneficiary of the 2021 Brain Prize for his pivotal examination finding the job of CGRP in headache, and co-creator of the stage 3 preventive investigation distributed in The Lancet, said the new sign for rimegepant is perhaps the most earth shattering things to occur in the 40 years that he has been rehearsing migraine medication.
“To have 1 drug patients can use to treat and forestall headache will probably change the treatment worldview for a large number of the large numbers of individuals who live with headache,” said Goadsby.
The new endorsement for rimegepant depends on a twofold visually impaired, randomized, fake treatment controlled stage 3 clinical preliminary with an open mark expansion. Essential examination endpoint results exhibited that rimegepant diminished month to month headache days by 4.3 days/month following 3 months of treatment, contrasted with fake treatment.
The preventive impacts of rimegepant were viewed as right on time as the main seven day stretch of treatment, as per Biohaven. Moreover, a key optional endpoint result showed that around half of patients treated with rimegepant had a half or more prominent decrease in the quantity of moderate-to-extreme headache days
___________________________________
FDA | Don’t forget to follow us on Twitter @njtimesofficial. To get latest updates