Bradley J. Priest, MD, FACS, FACOG, teacher, Division of Gynecologic Oncology, Arizona Oncology (US Oncology Network), University of Arizona College of Medicine, Creighton University School of Medicine at St. Joseph’s Hospital, clinical chief, Gynecologic Program, US Oncology Research Network, and co-chief, GOG Partners, talks about the utility of PARP inhibitors as support treatment in ovarian cancer.
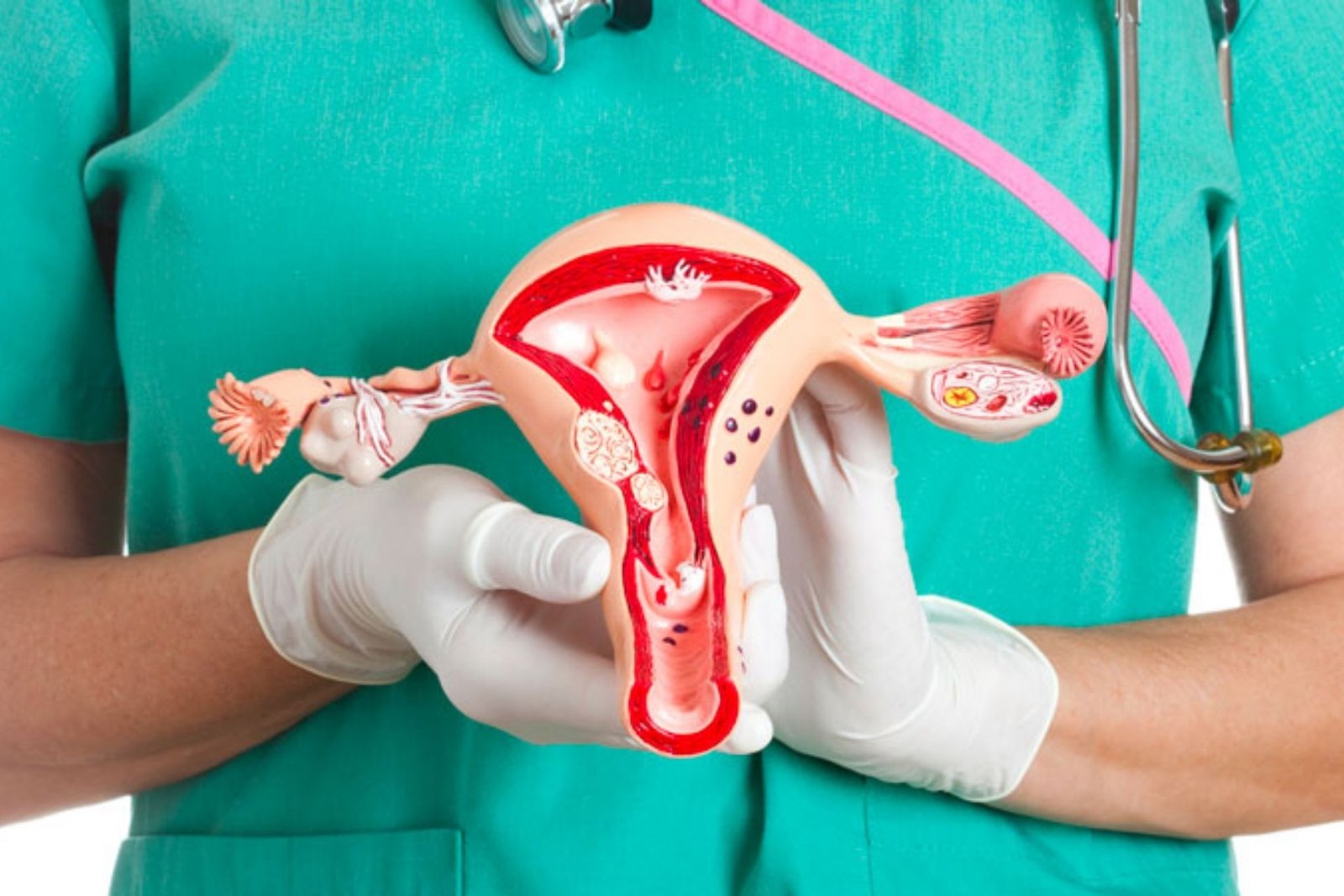
Of the 9 FDA endorsements for PARP inhibitors for use in ovarian malignant growth, 3 are shown for use as upkeep treatment in the repetitive setting for patients with platinum-delicate sickness, Monk clarifies.

These patients must have a reaction to platinum-based treatment to meet all requirements for upkeep PARP hindrance per the consideration rules from the stage 2 Study 19 (NCT00753545) preliminary, and the stage 3 SOLO-2 (NCT01874353), NOVA (NCT01847274), and ARIEL3 (NCT01968213) preliminaries.
Also, patients with platinum-touchy sickness are probably going to react to upkeep PARP restraint independent of homologous recombination insufficiency or BRCA status, Monk says.
Treatment choices are expected to choose between olaparib (Lynparza), niraparib (Zejula), and rucaparib (Rubraca) in light of the fact that the FDA signs are indistinguishable, Monk clarifies. The choice requires a common and customized approach between the patient and supplier, Monk finishes up.
_____________
PARP | Don’t forget to follow us on Twitter @njtimesofficial. To get the latest updates